MOBILE APPLICATIONS/WEB-APPLICATIONS / DESIGN/ ANALYTICS/MAINTENANCE
developers
120
years active
13
projects
522
Top-rated on Clutch among global developers
And top-10 among mobile developers according to AppFutura






WE WILL HELP YOU DESIGN YOUR OWN DIGITAL PRODUCT TO OPTIMIZE INTERNAL BUSINESS PROCESSES AND IMPROVE RELATIONSHIPS WITH CUSTOMERS.
There is a list of healthcare development services we are offering at the moment:
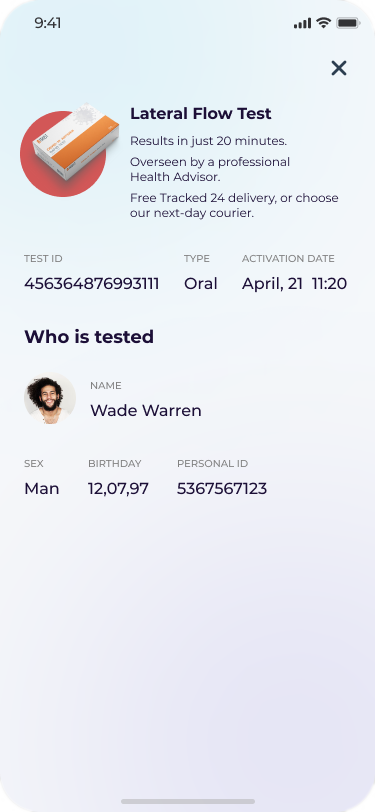
Health monitoring apps
Telemedicine apps
Reminders and alerts apps
Productivity apps
Patient data analysis apps
Customer relationship apps
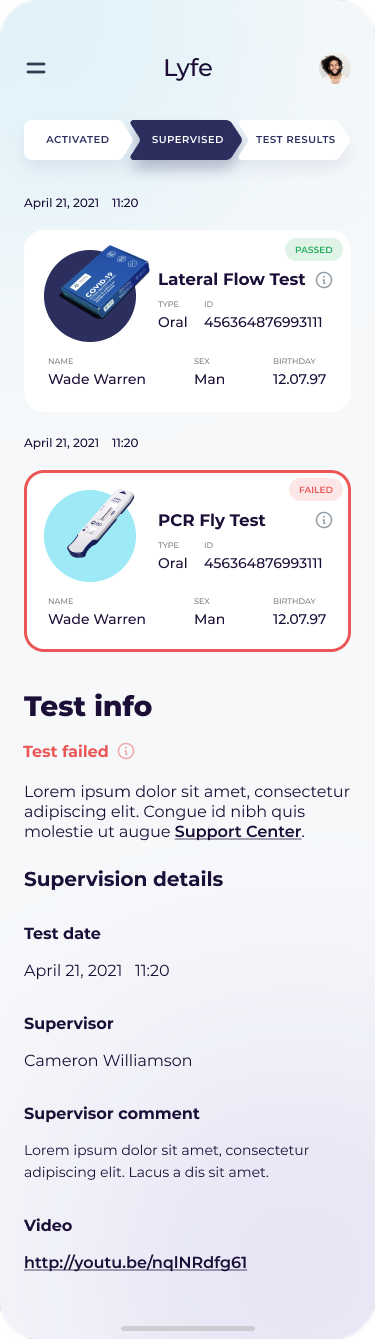
Medical records software
Hospital management software
Medical decision-making tools
Telemedicine apps
Reminders and alerts apps
Productivity apps
Patient data analysis apps
Customer relationship apps
Medical records software
Hospital management software
Medical decision-making tools
Magora guarantees a high level of expertise in Healthcare Software Development
Based on years of experience, we confirmed the adherence of our development services to the international standards in healthcare. We successfully integrate effective advanced technologies, thus increasing the potential of the whole industry.




Custom healthcare software development for every need
Magora takes an active role in the creation and modernization of medical software. We offer a comprehensive solution for a wide range of healthcare specialists, including health professionals, digital health and medtech companies, pharmacies, and hospitals. We also take care about knowledge transfer and create apps to provide students, patients, and practicing staff access to the most current and essential information. Monitoring apps are a great opportunity to control health indicators for the average person.

Pharmaceutical companies
We build and customize software solutions for drug development, clinical trials, production, and distribution. These include simulation and data visualization tools, ERP and supply chain management systems, and accounting software

Medical device manufacturers
Our team gives voice and processing power to hospital-grade and consumer devices. To that end, we develop embedded systems, mobile/web/cloud solutions for device management, and AI-driven data analytics tools

Healthcare providers
Our company develops EMR/EHR, practice and laboratory management, hospital asset tracking, remote patient monitoring (RPM), point of care (POC), and population health management (PHM) solutions

Biotech companies
We help biotechnology companies from the healthcare, pharma, agriculture, green tech, and industrial manufacturing sectors develop and deploy innovative biotech solutions — from laboratory software to organs-on-chips (OoCs).

B2B/B2C digital health startups
Partner with Magora to create telehealth and telemedicine systems, wellness technology solutions, including Femtech and Baby Tech products, and software infrastructure for diagnostic and assistive devices

Life science companies
Magora creates standalone apps and entire software ecosystems facilitating research and development (R&D) activities in the life science field. Whether you’re developing a new medication or biodegradable fuel, our life science consulting company is here to help!
The technologies we use
We responsibly approach the work and choose the best and most advanced technologies that save our clients’ time and projects’ costs, provide data security, and ensure smooth operation. Magora leverages all of its knowledge of the technical side of things with an understanding of the healthcare industry to bring an idea to realization.
Our Development Process
Discovery phase
Prototyping and MVP
Development and QA
Publishing and support
• Gathering and evaluation of requirements
• Revealing the business value and technical complexity that awaits the project
• Revealing the business value and technical complexity that awaits the project
• Establish a design concept
• Building main contract functionality
• Building main contract functionality
• Contract and test engineering
• The back-end and front-end development
• Testing: manual and automated
• External audit
• The back-end and front-end development
• Testing: manual and automated
• External audit
• Market launch and deployment of the mainnet
• Support after launching and the analysis of user acquisition
• Updates based on feedbacks a blockchain
• Support after launching and the analysis of user acquisition
• Updates based on feedbacks a blockchain




Our healthcare software development expertise
At Magora, we use effective tools to build custom software and apps for the healthcare industry while ensuring security against cybercrime and cyberattacks.
We know how to save patients’ data and your company’s IT infrastructure. Magora’s team creates stable software for healthcare needs, complying with all international regulations such as GDPR, DIGA, HIPAA, ISO 14971, ISO 13485.
We also value the importance and accessibility of information. Using Fast Healthcare Interoperability Resources (FHIR) we strive to ease the process of data exchange for our software development solutions. The standard advances interoperability, represents, and shares data among organizations in one standard way, regardless of its representation in the data storage.
We also value the importance and accessibility of information. Using Fast Healthcare Interoperability Resources (FHIR) we strive to ease the process of data exchange for our software development solutions. The standard advances interoperability, represents, and shares data among organizations in one standard way, regardless of its representation in the data storage.
Based on the standards DIGA, ISO 14971, ISO 13485 Magora is ready to develop the
software part to integrate your medical device.
We also value the importance and accessibility of information. Using Fast Healthcare
Interoperability Resources (FHIR), we strive to ease the process of data exchange for our
software development solutions. The standard advances the interoperability of the healthcare system, which means that information can be securely shared between electronic health records (EHRs), mobile apps, and organizations’ servers in one standard way, regardless of its representation in the data storage. It enables us to manipulate essential data easier than ever and results in cost-efficient care. Test results and treatment summaries are now easily accessible on your smartphone. Accessibility is one of the great benefits of FHIR, as it leads to a patient-centric approach. The technology implies a unique identifier that makes available necessary records, bills, and payments from any device or an application.
software part to integrate your medical device.
We also value the importance and accessibility of information. Using Fast Healthcare
Interoperability Resources (FHIR), we strive to ease the process of data exchange for our
software development solutions. The standard advances the interoperability of the healthcare system, which means that information can be securely shared between electronic health records (EHRs), mobile apps, and organizations’ servers in one standard way, regardless of its representation in the data storage. It enables us to manipulate essential data easier than ever and results in cost-efficient care. Test results and treatment summaries are now easily accessible on your smartphone. Accessibility is one of the great benefits of FHIR, as it leads to a patient-centric approach. The technology implies a unique identifier that makes available necessary records, bills, and payments from any device or an application.
We develop apps specifically tailored to meet your requirements
We develop apps for Medtech and Pharma companies.
We will analyse your pain points and find the best solution to increase your revenue and attract more clients.
We will analyse your pain points and find the best solution to increase your revenue and attract more clients.
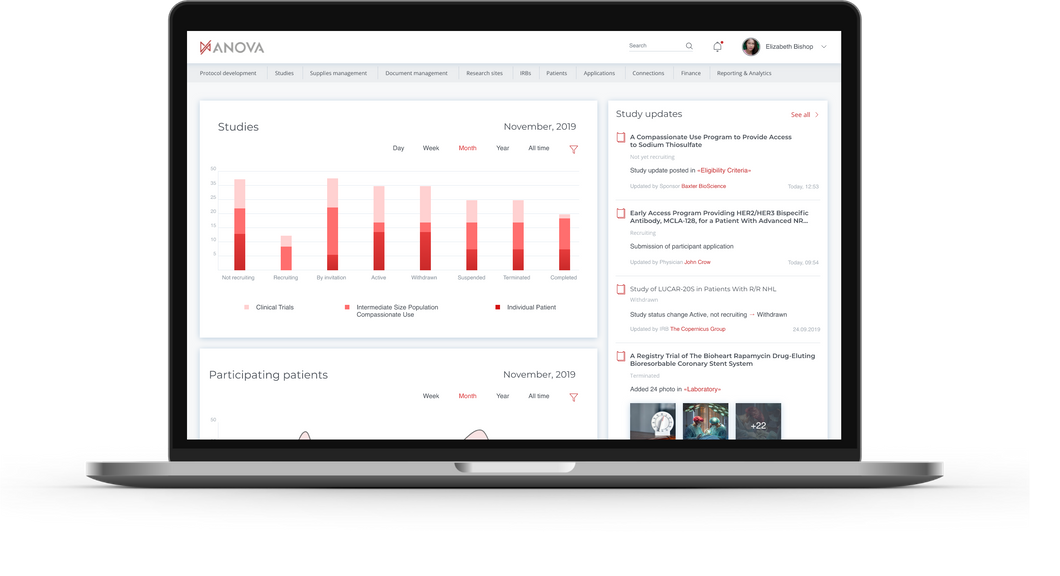

We designed a platform that automates clinical trials bringing Sites, Sponsors and Research Organizations together.
—platform that automates clinical trials
Anova
01.
01.

It exchanges the hundreds of Excel files each medical representative has to fill out, while providing clear statistics on product sales and the contract performance.
—enterprise software system for field sales personnel and management for Pharmaceutical company EGIS
PharmCard
02.
02.
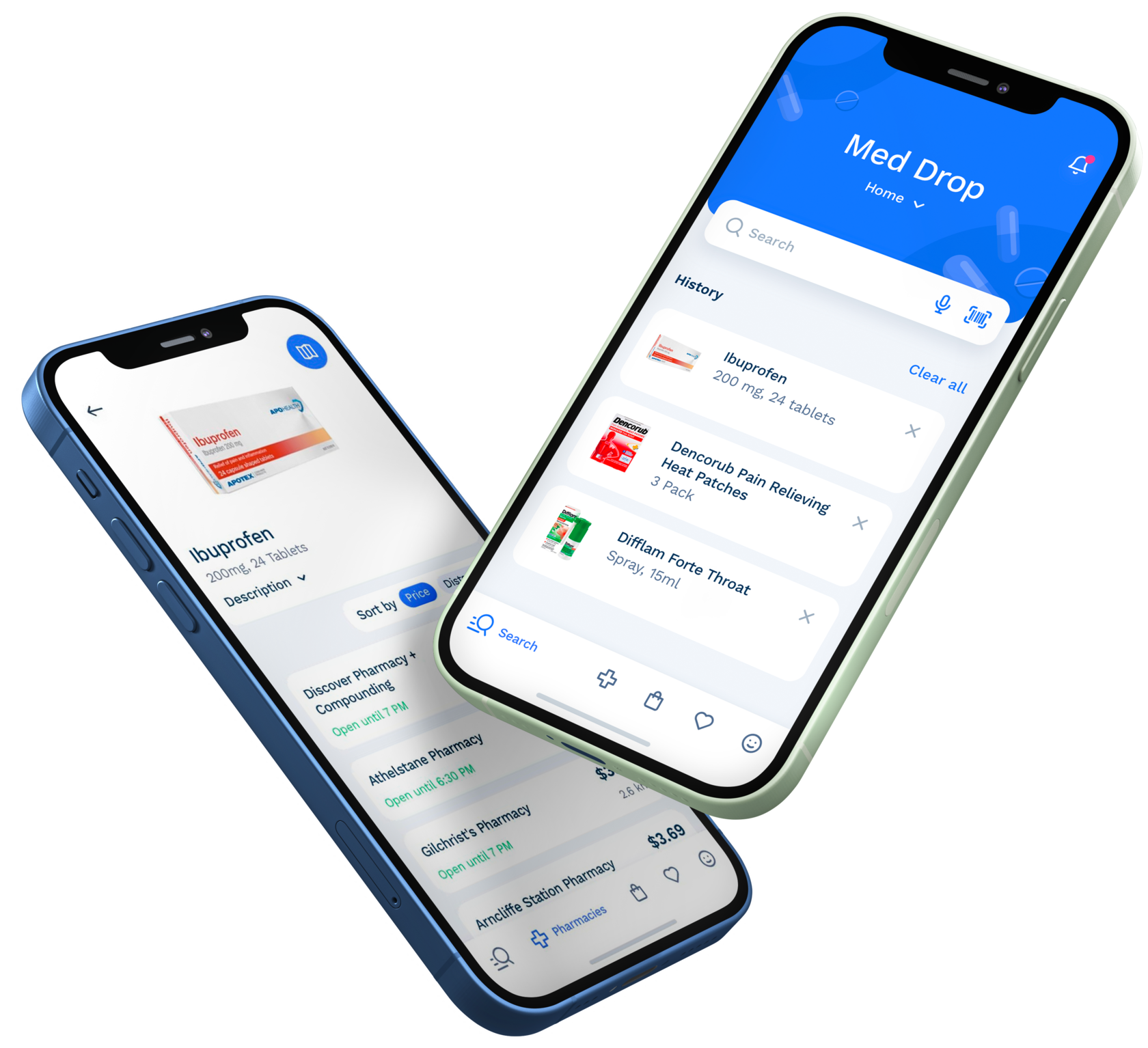

It allows to choose cheaper medicine and get it delivered much faster. It also helps track your order online with your mobile app.
—online pharmacy aggregator-platform that helps deliver medicine from pharmacies to end customer
MedDrop
03.
03.
Finding sponsorship can be time-consuming and expensive making the entire process more difficult, yet slowing down the research. No access for sponsors to packages regulation, streamlined submission and approval process can hamper development acceleration. The process of capturing qualified site and personnel profiles is very slow. Patient enrollment to clinical trials is not carried out on time.
Objective: design a web application that would bring labortories, sponsors and reseach organizations together to kickstart the development of new treatment through standartization and automation.
Objective: design a web application that would bring labortories, sponsors and reseach organizations together to kickstart the development of new treatment through standartization and automation.
All sponsors and active studies are captured globally. Protocols are structured and eligibility criteria codified to match to patient clinical characteristics. A direct access to sponsor program leads provided for rapid activation and enrollment. Qualified site and personnel profiles captured with all research interests, regulatory credentials and operational capabilities
Magora offered a very convenient soluton that is integrated with client's CRM.
It enables the EGIS managers to see relevant sales data and stocks online using a convenient web interface. With a mobile application medical representatives can enter data regarding quantity and sales volume online.
It enables the EGIS managers to see relevant sales data and stocks online using a convenient web interface. With a mobile application medical representatives can enter data regarding quantity and sales volume online.
- We developed a platform-aggregator for managers where they can track stocks, sales volume and сouriers performance.
- We developed a mobile application for couriers which gives them a full access to the information about new orders and each order payment.
- We developed a mobile application for clients that allows them to find the medicine they need at their location, order medicine and get it delivered to any destination.
- We added features allowing to scan bar-code, manage pharmacy stocks and aid patients in finding the medicine they need. We also added a feature allowing to scan Q-code to make the entire delivery process quick and smooth.
We provide a recognised healthcare development service for your great idea
We are trusted by big brands and emerging businesses, thus you can always count on our expertise if you want to make it big in the industry
Clutch
Software Suggest
AppFutura
Good Firms




5/5
5/5
5/5
5/5




Let’s talk and work together!